Following a report about patients receiving hydroxychloroquine dying at higher rates than other coronavirus patients, the World Health Organisation has temporarily suspended the trials of the anti-malarial drug as a possible cure for coronavirus.
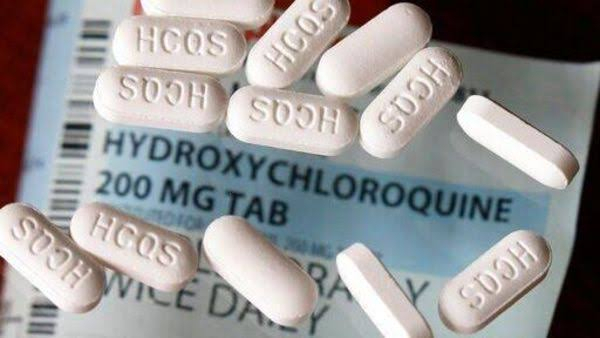
Despite medical and scientific experts warning against the consumption of hydroxychloroquine as a precaution or cure against coronavirus, the drug gained popularity after US President Donald Trump claimed to have taken it for nearly two weeks.
Lancet Report
A study published in The Lancet spoke about fatal effects of the anti-malarial drug being used to treat coronavirus patients. Confirming the suspension of the hydroxychloriquine coronavirus trial, WHO chief Tedros Adhanom Ghebreyesus said that the executive group of the so-called Solidarity Trial, which includes several hospitals in different countries enrolled for testing positive patients for possible COVID-19, has suspended trials using that drug, as a precautionary measure.
"The Executive Group has implemented a temporary pause of the hydroxychloroquine arm within the Solidarity Trial while the safety data is reviewed by the Data Safety Monitoring Board," he said.
The Daily Mail reported that the WHO chief confirmed that other arms of the trial are continuing, including testing of the experimental drug remdesivir and an HIV combination therapy.
Hydroxychloroquine Increase Heart-Related Complications - Study
The study published in The Lancet on Friday included over 96,000 people hospitalized with coronavirus. It included those treated with hydroxychloroquine and chloroquine.
The report stated: "We were unable to confirm a benefit of hydroxychloroquine or chloroquine, when used alone or with a macrolide, on in-hospital outcomes for COVID-19. Each of these drug regimens was associated with decreased in-hospital survival and an increased frequency of ventricular arrhythmias when used for treatment of COVID-19."
NPR quoted WHO's chief scientist, Soumya Swaminathan, stating that the suspension was prompted following the study. "While it was still a reporting of observational data it came from multiple registries and quite a large number of patients, 96,000 patients. The steering committee met over the weekend, in the light of this uncertainty. We decided we should be proactive, err on the side of caution and suspend enrollment temporarily into the hydroxychloroquine arm [of the Solidarity trial]," she said.
WHO Warns About Second Peak of Coronavirus
Mike Ryan, head of the WHO emergencies programme, warned about the possibility of the virus peaking again in the wake of the recent easing of lockdowns the world over.
"We need also to be cognizant of the fact that the disease can jump up at any time. We cannot make assumptions that just because the disease is on the way down now, it is going to keep going down and we get a number of months to get ready for a second wave. We may get a second peak in this wave," he said.
Since its outbreak in Wuhan province of China, in December last year, the global pandemic has infected over 5.5 million people globally and killed more than 347,000 people









