A baby boy in the U.K. became the first to get a cannabis-derived medicine for a trial to investigate whether the drug can prevent seizures and brain damage in newborns. The baby, Oscar Parodi was born via an emergency C-section on March 11.
Soon after his birth, he was transferred to the Neonatal Intensive Care Unit (NICU) at the Norfolk and Norwich University Hospital (NNUH), around 100 miles from London where one part of the vaccine trial is taking place.
The boy was given the cannabis-derived drug as part of the process to examine and understand whether the medicine is safe and effective in babies with Neonatal Hypoxic-ischemic Encephalopathy (HIE), which is thought to be the most common cause of seizures in babies and can cause the death of brain tissue. At this moment, there is no specific drug or treatment method to help HIE patients. This issue occurs when a fetus doesn't get enough blood and oxygen from the placenta.
Trial for Potential HIE Drug
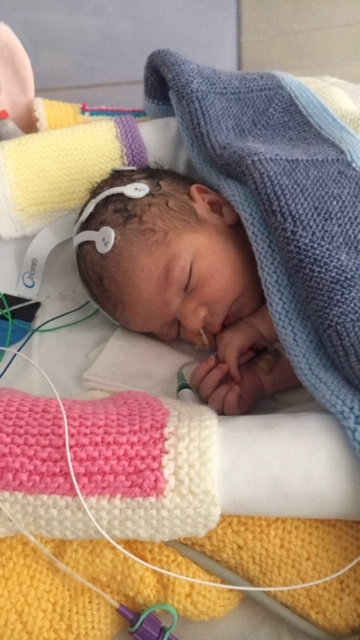
It should be noted that the team in the U.K. hospital which is running the trial, apart from Oscar Parodi, has recruited one other baby to the randomized trial. Newborns taking part in this trial will receive the standard treatment for HIE in which their bodies need to be kept at 33.5 degrees Celsius. As per the researchers, Oscar Parodi was given the cooling treatment for 72 hours.
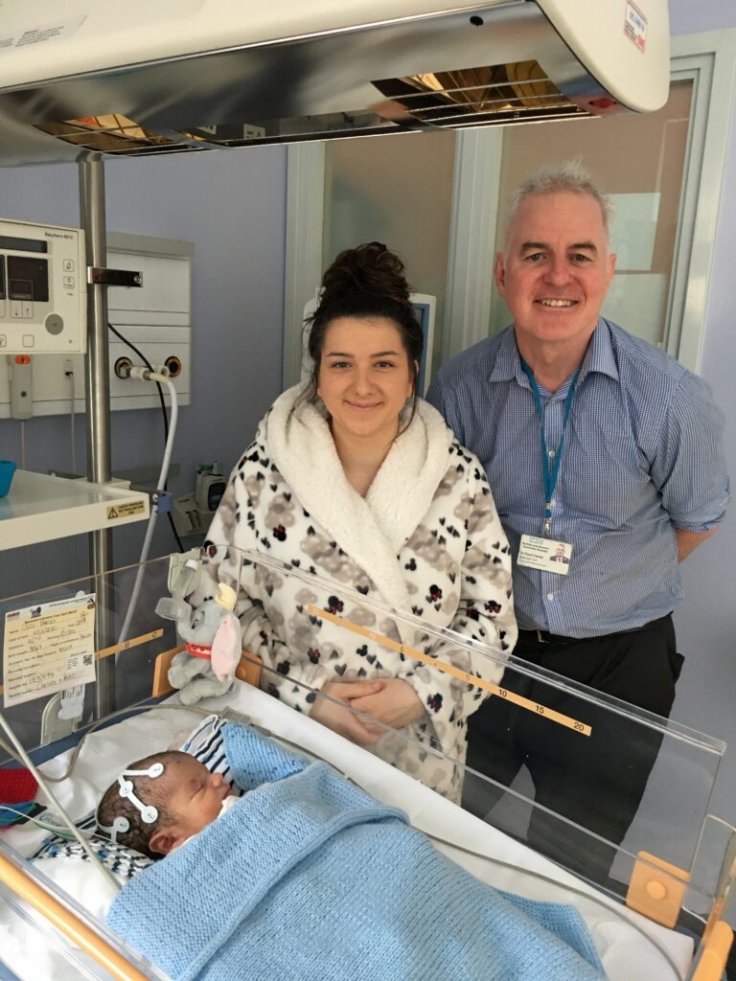
Oscar's mother Chelsea Parodi, of Watton, Norfolk, said, "I was approached after the birth about taking part in this study and I consulted my mum and my brother who is training to be a paramedic. It was hard but I wanted to do everything I could to help my baby boy.
"Oscar was in the hospital for nine days and he was being monitored 24/7. He is doing fantastically well and I am really grateful to Dr. Clarke and the team for what they have done for us."
As per the news release by NNUH, the babies will be randomly assigned either the cannabis-derived medicine or a placebo as soon as possible starting the hypothermia treatment and 12 hours after their birth. The researchers will collect their blood samples and measure the electrical signal in the brains of the babies. The follow-ups will be arranged with the families of these babies within the brackets of 30 days, six months, and 12 months period after they leave the hospital.
It should be mentioned that the medication will be given to the babies through their veins or intravenously which is thought to be the world's first. They will be given a 30th dose of the drug which is developed to ensure that it contains a minimum level of tetrahydrocannabinol (THC), an ingredient that makes the cannabis users high.
Research in the UK
Professor Paul Clarke, consultant neonatologist at NNUH, said that it is the first time a cannabis-derived drug has been tested intravenously in human babies. In addition, he said, "One of the attractions of this trial for parents is the closer brain monitoring that babies get as part of the study because a more advanced brain wave monitor is used for the trial babies. This gives parents more reassurance that any seizures will be picked up."
He mentioned that there may be unexpected side effects and unknown risks, so keeping this in mind, "The trial has been carefully designed to make it as safe as possible and so we are only giving the babies a minuscule dose at the beginning and we monitor them even more closely than usual."









