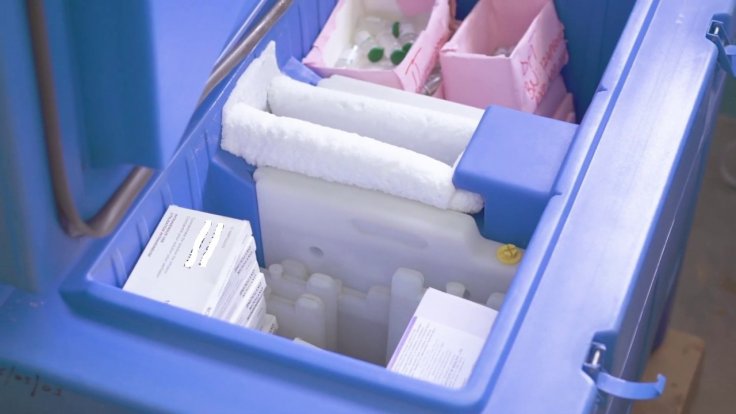
Thousands of doses of the Covid-19 vaccine had to be returned to Pfizer over the weekend after an "anomaly" in the transportation process caused storage temperature to drop below the required level, authorities said on Wednesday. The doses were en route to California and Alabama and were closely monitored but still for some unknown reason the temperature in the storage boxes abruptly dropped.
This once again brings forth the several challenges involved in storing and shipping the Pfizer-BioNTech Covid-19 vaccine and raises concern over the smooth distribution of the doses in different parts of the world. It is still unclear what caused the storage temperature to fall below the special level.
First Setback

As many as four trays containing the Pfizer-BioNTech Covid-19 vaccine, which despite being closely monitored while being transported to California and Alabama, witnessed a sudden drop in temperature from minus 80 degrees Celsius to minus 92 degrees, according to Gen. Gustave Perna, the head of Operation Warp Speed. The vials of the vaccines are stored in trays, each carrying 975 doses.
Pfizer's Covid-19 vaccine needs to be stored in ultra-cold freezers at minus 70 degrees Celsius. However, it's still unclear how the temperature dropped suddenly while in transit. Interestingly, the same incident happened at two different locations. The first two trays were headed for California, when the anomaly was detected. Similar reports came from Alabama few hours later where two more trays were affected.
"We locked those trays down," said Perna at a press briefing. "They never left the truck. We returned them immediately to Pfizer and we sent immediate shipments to replace those two trays."
Pfizer's Many Challenges
Authorities are now working with the Food and Drug Administration (FDA) and Centers for Disease Control along with Pfizer to determine if such anomaly is safe in the future. However, these doses won't be used as authorizes wasn't to avoid all kinds of risk.
A Pfizer spokesperson issued a statement saying: "The returned shipments, totaling 3000 doses, will be reviewed internally by Pfizer following our quality investigative process, however, it's Pfizer's vision that its customers should not have to wait while these reviews take place."
Pfizer's Covid-19 vaccine was the first to win emergency authorization from the FDA and immunizations began this week. Operation Warp Speed has said it expects to have enough vaccines between Pfizer and another shot on deck from Moderna to inoculate 20 million Americans in December.
However, Pfizer has already sighted production challenges which might show down the inoculation process. Nearly two million doses of the vaccine will be allocated to the United States next week, less than the 2.9 million available this week when the first shots shipped, authorities said without citing any specific reason behind fewer doses being rolled out.









