Heart diseases or Cardiovascular diseases (CVDs) is one of the leading causes of death across the world. This silent killer creeps up unsuspecting individuals and announces its domination often in the form of myocardial infarction (MI) or a heart attack. MI leaves the heart acutely damaged. However, a new study reports the development of a cardiac patch that can help in the recovery of damage caused to heart tissue.
In the study, published in the journal ACS Biomaterials Science & Engineering, scientists described the development of a patch that integrates biomimetic microvessels (BMVs) and cardiosphere-derived stromal cells (CSCs) in order to repair ischemic (poor supply of blood) damage, and showed success in rat and pig models. "These findings establish the BMV–CSC patch as a novel engineered-tissue therapeutic for ischemic tissue repair," the authors wrote.
A Global Killer

According to the World Health Organization, CVDs caused the death of approximately 17.9 million people worldwide in 2016—31 percent of all deaths in the world. 85 percent of these deaths were caused by MI or heart attack.
MI is caused when the artery/s is clogged by a blood clot that prevents the flow of blood to the heart. Without receiving sufficient blood, the tissue loses oxygen. This in turn causes extensive death of cardiac cells, harming of blood vessels and inflammation. The effective treatment of MI is possible only when heart muscle tissue can regenerate and new blood vessels are formed to restore nutrient and oxygen supply to the cells.
A Patch to Mend Hearts
Corresponding author Ke Chang and his collaborators had previously designed a relatively easy-to-manufacture pre-vascularized (adding blood vessels) cardiac patch. It contained engineered microvessels(synthetic blood vessels) suspended in a gel composed of the protein, fibrin, and induced with connective tissue cells known as cardiac stromal cells.
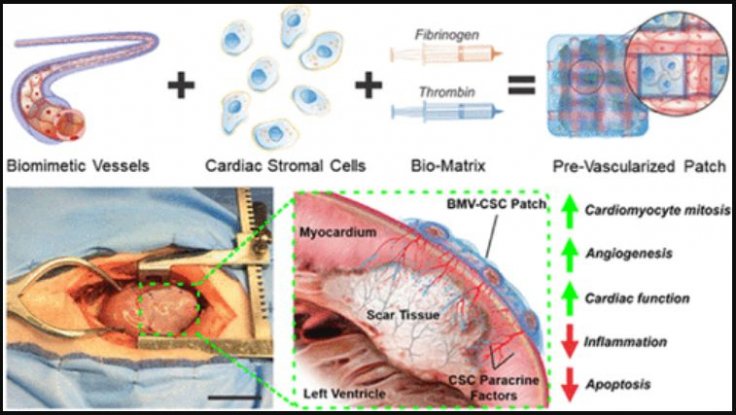
When the rats who had suffered from MI were implanted with the patch, the cells loaded in the patch secreted growth factors that aided in the regeneration of cardiac muscle and blood vessels. Now, the authors wanted to test the effectiveness of the patch in rats further. However, they also sought to test its performance in the hearts of pigs as their cardiovascular system bears more similarities with humans than rodents.
More than Promising Results
Rats that suffered MI recently were implanted with the novel patch. Four weeks later, the authors noticed that the rats that had received the patch showed lesser scar tissue, more cardiac muscle, and enhanced pumping function in comparison to rats that had not received the patch for treatment.
When pigs that had experienced MI were implanted with the patch, the scientists found that they also had undergone similar changes. Not only did the patch heighten the recruitment of progenitor cells in the pigs to the site of damage, but it also improved the growth of new blood vessels. It also reduced the death of cardiac cells and was able to suppress inflammation.

"We showed that BMV–CSC patch transplantation significantly promoted cardiac function, reduced scar size, increased viable myocardial tissue, promoted neovascularization, and suppressed inflammation in rat and porcine MI models, demonstrating enhanced therapeutic efficacy compared to conventional cardiac stromal cell patches," the authors wrote.
First of Its Kind
It is important to note that research into the development of cardiac patches has been ongoing and has employed used natural blood vessels or blood vessel-forming cells to vascularize them. However, the current study holds the distinction of being the first to utilize the pre-vascularized cardiac stromal cell patches using micro-engineered synthetic blood vessels for treating MI and successfully demonstrating its effectiveness.
Additionally, the study is also the first to prove the efficiency of such a patch in a large animal model. The authors stressed that the mechanisms, safety, and efficacy of patch repair require further study in order to make sure that that technology can be safely applied to humans.









