For patients suffering from cancer, the impact of treatments is not always uniform and effective. Treatments such as cancer immunotherapy and chemotherapy may not be effective in all the patients and the chances of failure are higher. However, offering hope to sufferers of pancreatic cancer, a new study has described the development of a simple experimental blood test that can tell which form of the cancer is treatable and which are not.
According to the authors, such a critical distinction can enable oncologists in making vital decisions that spare patients ailing with resistant cancers from subjecting themselves to unhelpful treatments that can lead to side effects and adding to their distress.

"Knowing which type of pancreatic cancer a person has is critical to implementing the right treatment strategy for each patient. We hope that our new test, which detects a marker produced by cancer cells of one subtype and not the other, will one day soon be a powerful tool to help physicians and patients make the best decisions possible," Dr. Brian Haab, corresponding author of the study, in a statement.
A Tricky Cancer to Diagnose
According to the World Cancer Research Fund, pancreatic cancer is the 12th most common cancer affecting men, and the 11th most common one among women. There were nearly 460,000 new cases in 2018 alone. The total number of deaths owing to pancreatic cancer was over 432,000. What makes the cancers of the pancreas some of the most difficult malignant cancers to treat is their ability to evade detection at an early stage, and are often diagnosed at an advanced stage.
Currently, oncologists do not have a reliable method of learning whether their patients have a subtype that may not respond well to treatments in comparison to subtypes that may. Thus, most patients receive a standard treatment approach that may be effective in some but may be ineffective in others and cause side effects.
Detecting Treatable Pancreatic Cancer Using A Blood Test
The new experimental test is based on the detection and measuring of the levels of a sugar known as sTRA. This sugar is produced in patients with certain subtypes of pancreatic cancer and enters their bloodstream. In those pancreatic cancers where sTRA is produced, chemotherapy mostly does little to improve the condition.
The new test is developed from an older sTRA test published by Haab and his team in January 2019. That test also demonstrated an experimental test of the blood that was a combination of an existing diagnostic that detects a sugar known as CA19-9 with the new test that specifically detected sTRA.
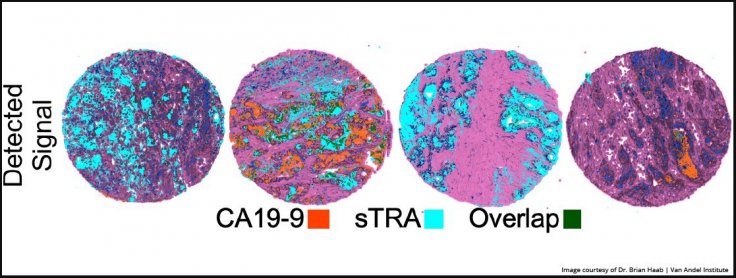
The combined approach was able to detect nearly 70 percent of pancreatic cancers with a false-positive rate of less than 5 percent—which is roughly 30 percent higher than the use of CA19-9 alone. Both the tests—the new test and the one from 2019—are set to undergo further clinical validation.
"The 2019 combination test tells us whether there is cancer and the new sTRA test helps us determine what kind of pancreatic cancer, which then could allow physicians to better narrow down the appropriate treatment plan. When used in sequence, we believe the combination test and the new sTRA test could help catch and identify pancreatic cancer more quickly and definitively," concluded Dr. Haab.









