The onslaught of the SARS-CoV-2 coronavirus has been an unpredictable one. While some have been lucky enough to contract mild forms of COVID-19 without any serious damage, some have been hit by severe forms that have led to adverse outcomes such as multiple organ failure, and ultimately death. Now, in a new study, researchers have managed to discover how the novel coronavirus damages several key organs, including the heart and kidneys.
Scientists from the University of California - Los Angeles (UCLA) created a variety of COVID-19 in a mice model that revealed how the notorious virus damages multiple organs in the body by disrupting the production of energy in their cells and causing genetic changes.
"This mouse model is a really powerful tool for studying SARS-CoV-2 in a living system. Understanding how this virus can hijack our cells might eventually lead to new ways to prevent or treat the organ failure that can accompany COVID-19 in humans," said Dr. Arjun Deb, co-senior author of the study, in a statement.
Making Mice Human-like
Several studies have found that the SARS-CoV-2 virus gains entry into human cells by binding with a protein known as angiotensin-converting enzyme 2 (ACE2) receptors that are found on the surface of cells. The ACE2 receptors found in mice, however, are not recognized by the virus. Thus, upon being exposed to the pathogen, healthy mice are not affected by it.
Therefore, in order to study the human effects of the virus in mice, the rodents have been engineered for several studies to possess the human variant of ACE2 in their lungs, and then infected with SARS-CoV-2 through their nose. Despite being infected and exhibiting some of the symptoms such as pneumonia, the mice generally do not manifest a wide range of symptoms as human beings do.
COVID-19 studies in humans have suggested that the virus can reach multiple organs by traversing through the bloodstream. Dr. Deb pointed out: "Among COVID-19 patients, those who have organs involved other than the lungs are most at risk of a bad outcome." Hence, the current study focused on making mice contain human ACE2 in key organs other than just the lungs and learning how the novel coronavirus harms other organs
Infecting Rodents to Understand Organ Damage

For the study, the team engineered mice that possessed a human version of the ACE2 receptor not only in the lungs, but also in other vital organs such as the heart, spleen, and kidneys, among others. Next, they infected half the test animals with the SARS-CoV-2 virus by injecting it directly into their bloodstreams. The scientist monitored the overall health of the mice over the following days and examined the change in the levels of certain proteins and genes in their bodies.
The infected mice stopped consuming food within seven days. They also displayed complete inactivity and weight loss that amounted to around 20 percent of their body weight. On the other hand, the mice that had been designed to contain human ACE2 but were not infected by the researchers did not show notable weight loss.
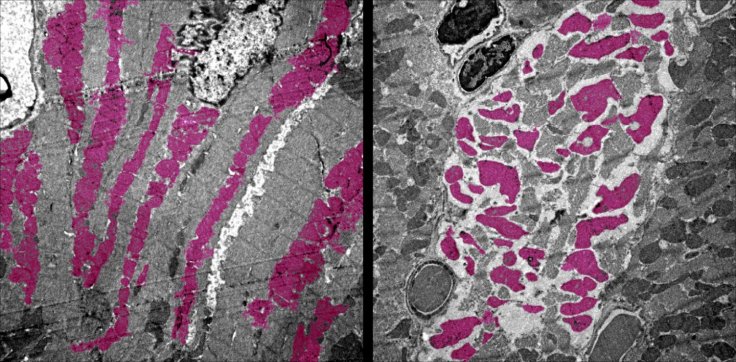
Additionally, the levels of immune cells in the infected animals were found altered. Most importantly, they exhibited serious human symptoms observed in patients who are severely ill with the novel coronavirus infection, such as the spleen wasting away and swelling of heart tissue.
Virus-induced Alterations to Genes
Other than the visible damage that the SARS-CoV-2 virus caused, the team also investigated the effect it had on a genetic level. The authors sought to ascertain which genes were switched on and off in the animals that had been infected with the disease. A startling discovery was made.
The tricarboxylic acid cycle, or TCA cycle, and electron transport chain—regular molecular processes through which cells produce energy—were shut down in the lungs, heart, spleen, and kidneys. "If a virus snuffs out the energy-generating pathways in multiple organs of the body, that's going to really wreak havoc," emphasized Dr. Deb.
However, the ultimate finding was that several changes that the organs of infected mice sustained were permanent or enduring. While the genes underwent alteration that let their switching on or off in certain cells, the SARS-CoV-2 caused epigenetic changes—observable chemical changes to DNA that lead to observable physical modifications—as well.
According to Dr. Deb, this could serve as an explanation for the persistence of symptoms in some COVID-19 patients for weeks and months even after recovering from the infection. He also added that while the study may not have an instant impact on the treatment of COVID-19, it can definitely aid in the trials of novel drugs for the treatment of the viral infection.









