The University of Washington estimated that half a million Americans will likely die of the Coronavirus in the US by April 2021, and if the mask mandate is relaxed, the total may rise to 770,000. Such numbers are scary as the US has lost more lives compared to any other COVID-19 affected nation. But here is a small hope.
Doctors in Tampa Bay, Florida are now using a new COVID-19 drug to keep patients out of hospitals. The healthcare professionals have claimed that the antibody cocktail from Eli Lily, known as bamlanivimab, is helping treat recently-diagnosed patients who are at high-risk.
Another antiviral drug, MK-4482 / EIDD-2801 or Molnupiravir, can suppress "completely" the transmission of the Coronavirus infection in just 24 hours, revealed a study conducted by the Institute of Biomedical Sciences of Georgia State University. The researchers said, "This is the first demonstration of an orally available drug that quickly blocks the transmission of SARS-CoV-2, so it could be a game-changer."
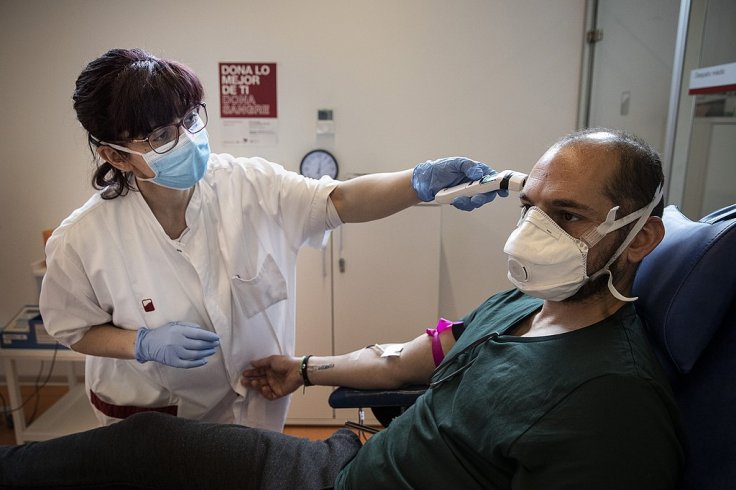
The Drug Used by Florida Doctors
Dr. Hal Escowitz, the Chief Quality Officer at Lakeland Regional Health said that the treatment involving the antibody cocktail is different from any other therapies that "we've had in the past in that this is the first drug [bamlanivimab] that we've had that is meant for outpatients." He explained that it is an IV therapy and "it's not for people once they're sick enough to be in the hospital. That actually excludes them from getting a drug."
Lakeland Regional Health has Florida's largest supply of bamlanivimab and as reported out of 38 patients who were treated at the hospital, as of now only one has been admitted. As per Dr. Escowtiz, "I think this is potentially a very lifesaving treatment."
One of the patients, 60-year-old veteran Thomas Sheridan, credited the drug for saving his life. He tested positive for the Coronavirus caused disease on November 24, and started experiencing body aches, headaches, fever, and more.
He went to the Lakeland Regional Health for the treatment and after receiving the therapy which took almost an hour to administer, Sheridan said it saved his life. While some people reported headaches as a side effect of the treatment, the elderly man in Florida said that the infusion left a bruise on his arm but no other side effects.
As per the Food and Drug Administration (FDA), "It is recommended that bamlanivimab be administered as soon as possible after the positive viral test for SARS-CoV-2 and within 10 days of symptom onset. Bamlanivimab is administered as a single dose of 700 mg via IV infusion over 60 minutes."
What About Molnupiravir?
This drug was developed at Emory University in Atlanta by Drug Innovation Ventures at Emory (DRIVE). The researchers said that tests were carried out on ferrets and it was noticed that they presented a reduction in the number of viral particles. Those ferrets were later put with others that had not been treated. According to the researchers, none of the ferrets from the second group became infected with novel Coronavirus.
The researchers believe that if the ferret-based data is translated into humans, novel Coronavirus infected patients treated with Molnupiravir could become non-infectious within 24 hours of starting treatment.








