The U.S. Food and Drug Administration (FDA) on Monday warned against using a combination of anti-malaria drug hydroxychloroquine and Gilead's experimental remdesivir on Covid-19 patients, as it could substantially reduce the effectiveness of the drug. The announcement came hours after the FDA revoked the emergency use of hydroxychloroquine as a potential Covid-19 treatment.
The FDA had earlier warned against prescribing malaria drugs chloroquine and hydroxychloroquine outside hospitals to treat Covid-19 after "serious" poisoning and deaths were reported. The drug was championed by President Donald Trump as a potential treatment for coronavirus but several studies showed that it was not effective to treat the novel coronavirus.
Combination Weakens Remdesivir's Effectiveness
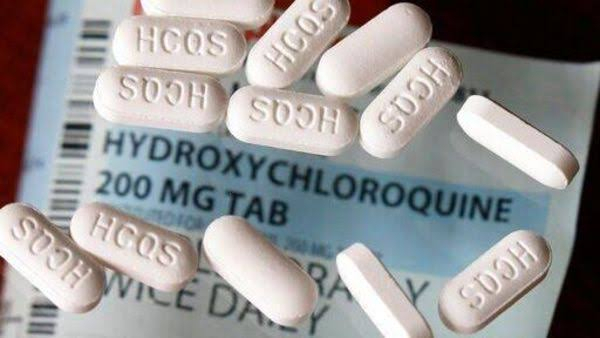
The FDA said that it is revising the fact sheet for healthcare providers that accompanied Gilead's experimental Covid-19 drug remdesivir to state that "co-administration of remdesivir and chloroquine phosphate or hydroxychloroquine sulfate is not recommended as it may result in reduced antiviral activity of remdesivir." Although FDA is still not confirmed if the effectiveness of remdesivir reduces if it is given in combination with hydroxychloroquine, it is evaluating all possible data available related to remdesivir.
"As we learn more about these products, changes may be necessary based on new data – such as today's updates for health care providers about a potential drug interaction and other important information about using remdesivir to treat COVID-19 patients," Patrizia Cavazzoni, acting director of FDA's Center for Drug Evaluation and Research said. Remdesivir now remains the only drug with an emergency use authorization from the FDA to treat Covid-19 patients in the United States.
FDA Stops Use of Hydroxychloroquine
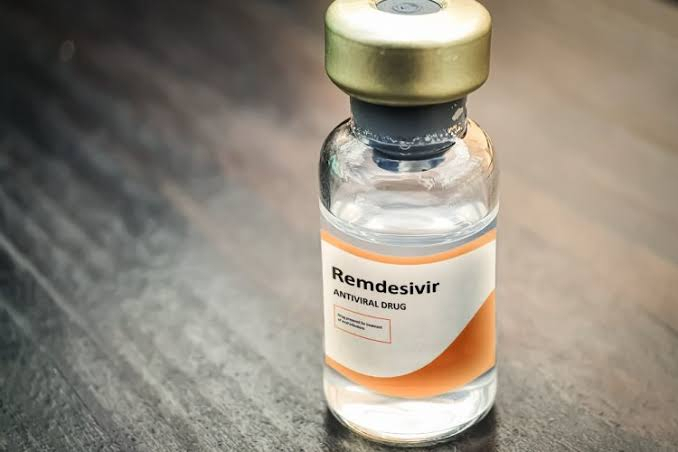
The announcement came hours after FDA revoked the emergency use authorization for hydroxychloroquine to treat the coronavirus. The FDA said that it is now affecting some doctors who had been prescribing it to Covid-19 patients and is unlikely to be effective. The FDA has also raised concerns over some possible side-effects of the drug and said that the known and potential benefits of chloroquine and hydroxychloroquine "no longer outweigh the known and potential risks."
In April, the FDA warned against using chloroquine and hydroxychloroquine outside hospitals after it learned about the increased use of drugs through outpatient prescriptions. The FDA had said that it learned of "serious heart rhythmic problems" in patients with Covod-19 infection who were treated with anti-malarial drugs hydroxychloroquine or chloroquine in combination with antibiotic azithromycin, commonly known as a Z-Pak.
Trump's Claims Proved Wrong
Chloroquine and hydroxychloroquine came to prominence in recent times after a small study on 36 patients in France in March found that most administered with the drug cleared the coronavirus from their system a lot faster than others. Since then Trump has touted it as a "game changer" in the fight against coronavirus.
However, a new study in the New England Journal of Medicine found that the drugs are nothing better than a placebo in preventing the coronavirus infection. The effectiveness of hydroxychloroquine and chloroquine was always questioned by researchers but Trump continued to push for the drugs calling it a promising tool to treat and prevent coronavirus. He even said last month that he had taken a dose of hydroxychloroquine as a safeguard.









