LabCorp's COVID-19 test kit is now the first non-prescription and over-the-counter product to receive an emergency use authorization by the US Food and Drug Administration (FDA) for at-home use.
Dr. Brian Caveney, chief medical officer and president of LabCorp, which is one of the largest diagnostics testing companies, said the first over-the-counter at-home collection kit ever authorized by the FDA for the Coronavirus caused disease will empower people to "learn about their health and make confident decisions".
After the approval announcement by FDA, Caveney also said that now the company can help more people to get tested, reduce the spread of the SARS-CoV-2 infection, and improve the health of people in the US, which has reported over 15 million cases and more than 289,000 deaths.
A Useful Home Test Kit
This newly approved LabCorp Pixel COVID-19 Test Home Collection Kit will allow anyone aged 18 or above to collect the nasal swab samples at home and then it will be sent to a LabCorp facility for testing. The kit users will receive positive or invalid results on phone or through a healthcare provider. If it comes negative, the users will be notified by email or through an online portal.
Dr. Jeff Shuren, the director of the FDA's Center for Devices and Radiological Health, said in the statement that many home collection kits can be prescribed with an online questionnaire. He said, however, this this newly authorized direct-to-consumer collection kit removes that step from the process, "allowing anyone to collect their sample and send it to the lab for processing". According to the FDA, this kit can help people understand their COVID-19 status and make decisions about when to quarantine.
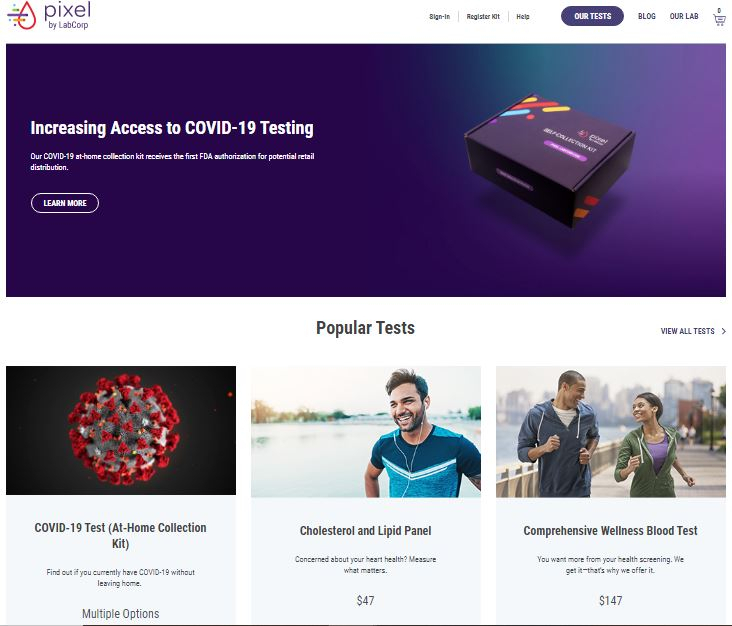
The kit is now available on Pixel by LabCorp website and is expected to be available in stores soon. When the consumers purchase it, they register the kit on the company's website and then have to follow the instructions given there.
In November, FDA granted the emergency use of the first rapid Coronavirus test—a 30-minute test kit from Lucira Health. This test kit is only available by prescription. FDA said that the 30 minutes test allows users to collect a nasal sample. The collected samples need to be put in a vial that plugs into a portable device that interprets the results and shows whether the person tested positive or negative for the Coronavirus caused disease.









