A defect in signalling pathway is responsible for a congenital heart condition known as left ventricular non-compaction (LVNC) that occurs when muscly projections in the embryonic heart fail to transform into compact heart muscle, scientists have found in a new study.
Scientists from Duke-NUS Medical School in Singapore said the new research could pave the way towards potential diagnosis and therapies for LVNC, in which an embryo develops a thinned lower left heart chamber, and reveal more information about the processes that regulate heart development.
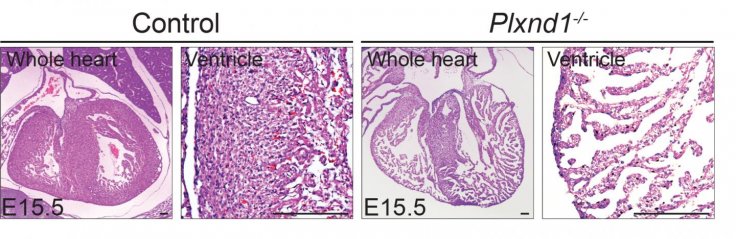
The finding published in the journal JCI Insight suggested that muscle cells migrate and proliferate, forming finger-like protrusions inside the cardiac chamber during a normal embryonic heart development, but these projections or trabeculae, which eventually dissolve, fail to dissolve and form solid heart wall leading to LVNC, one of the most common disorders in cardiac chamber maturation.
These trabeculae help with heart nutrition and oxygenation until blood vessels form, and their dissolution leads to the formation of a compact, smooth and solid heart wall.
"People born with LVNC often do not experience any symptom but are at a higher risk of heart failure, irregular heart rhythms, and cardiac arrest," said lead author and Assistant Professor Manvendra Singh from Duke-NUS' Cardiovascular and Metabolic Disorders (CVMD) Programme.
Singh and his colleagues made the discovery as they wanted to test if protein Sema3E -- known to connect with a cell receptor called PlexinD1, which is involved in embryonic heart development -- was linked to the defects seen in LVNC. Sema3E's existence in the heart, however, has not yet been determined.
Researchers found turning off Plxnd1 in preclinical embryonic heart led to increased activity from other genes initiated by a 'Notch signalling pathway', and inhibiting this particular pathway partially prevented the hearts from developing malformations.
"Our study helped us better understand the role of Semaphorin-Plexin signalling in this particular congenital cardiac condition," said co-author Reddemma Sandireddy, a Research Assistant in the CVMD Programme.
"Sema3E connects with its PlexinD1 receptor to turn off the Notch signalling pathway, thus suggesting its role in the formation and maturation of the lower heart chambers," she added.
"Every third death in Singapore is due to cardiovascular diseases, with Cardiomyopathy (heart muscle disease) being one of the most serious CVD. This research may provide potential new targets to treat the condition," said Professor Patrick Casey, Senior Vice Dean for Research at Duke-NUS Medical School.








