The number of COVID-19 cases reported and the increasing death toll due to the infection have left nearly 90 countries in crisis. According to the latest data, nearly 100,000 cases and over 3,200 deaths have been reported worldwide. As of now, there is no cure or vaccination for the disease caused by the new SARS-CoV-2 coronavirus, with scientists from across the world working around the clock to develop both.
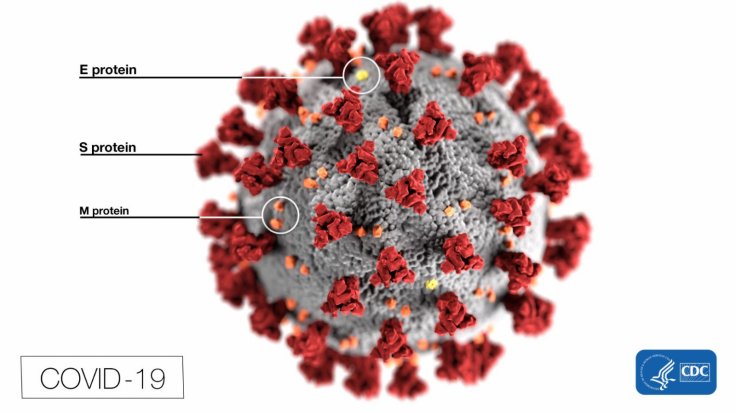
The SARS-CoV-2 is an enveloped, positive-sense, single-stranded RNA β-coronavirus that shares a lot of genetic similarities with other deadly coronaviruses such as Severe acute respiratory syndrome (SARS) and the Middle East respiratory syndrome (MERS). It is said to be over 80 percent identical to the SARS virus.
So an important question that arises is whether the treatment administered to treat the pathogen that caused the worldwide SARS outbreak in 2003 be used to deal with the current coronavirus outbreak. Also, can existing drugs be used to treat the infection?
How was SARS treated?
As of now, there are no standard or decisive protocols form the treatment of SARS. However, several regimens have been experimented with to varying degrees of success. Patients who are confirmed or suspected cases should be administered treatment that is normally used to treat any critical form of pneumonia, The Centers for Disease Control and Prevention(CDC) recommends.
The general pharmacotherapeutic approach towards the treatment of SARS has been in the use of cocktails antiviral drugs and corticosteroids, with ribavirin being used in Canada, Hong Kong, and other countries in the initial patients. However, subsequent reports suggested that ribavirin had no significant action against the coronavirus and in fact led to adverse effects such as hemolysis, the rupturing of blood cells.
Antiretroviral protease inhibitors and interferons were also used to treat the condition with diverse levels of efficiency. However, several drugs that were being developed against the disease saw no or limited development after the outbreak subsided.
Re-purposing of existing drugs to combat COVID-19
While several pharmaceutical companies and research facilities are fighting against time to discover a cure and a vaccine for the disease, attempts to re-purpose existing drugs to resolve the need is also on the rise. Since the beginning of the year, Chinese pharma companies have announced plans for several trials to test existing drugs for the treatment of COVID-19.
Ascletis Pharma, an Hangzhou-based company, sought the approval of Chinese authorities to test ritonavir and ASC09—protease inhibitors used to treat HIV—in trials. Another Chinese company, BrightGene Bio-Medical Technology, announced that it would start producing Remdesivir, a broad-spectrum antiviral agent manufactured by American pharmaceutical company, Gilead Sciences, for the treatment of COVID-19. Remdesivir was initially designed to tackle the Ebola virus.
With several genetic similarities between the three coronaviruses, and the availability of these drugs on a large scale, it is logical to turn to these existing drugs for a solution. "The general genomic layout and the general replication kinetics and the biology of the MERS, SARS and [SARS-CoV-2] viruses are very similar, so testing drugs which target relatively generic parts of these coronaviruses is a logical step," said Vincent Munster, chief, Viral Ecology Unit, US National Institute of Health, according to Nature.
Choosing the right drugs
Some of the antiviral targets encoded in the viral genomes of the above-mentioned coronaviruses include structural proteins, non-structural proteins and accessory proteins. While some play a vital role in their potency enhancing replication mechanism, others do not. Therefore, it is essential to identify the drugs that can be directed towards the genetic components of the COVID-19 that enhance its virulence.
Lopinavir/Ritonavir, an antiretroviral therapy, sold under the name Kaletra, is believed to inhibit a vital enzyme in SARS and MERS coronaviruses and was found to show promising results in trials against SARS.
However, Erik De Clercq, of the Rega Institute for Medical Research in Leuven, Belgium, said, "We should stay away from antivirals known to be acting at targets not playing a role in the replication of coronaviruses." He favours drugs that can target specific proteins such as the RNA-dependent RNA polymerase—an enzyme important in replication of the mentioned pathogens—of the SARS-CoV-2. This because unlike retroviruses such as HIV, coronaviruses lack or do not employ reverse transcriptase, an enzyme that converts the RNA into DNA.
George Painter, president of the Emory Institute for Drug Development, Emory University, also shares similar views of the protease targeting approach. He said, "It's probably a long shot to go for drug repurposing activity against the coronavirus using HIV drugs; these were protease inhibitors that were designed specifically for HIV." Nevertheless, there are several trials for Kaletra, alone or in combination with other drugs, that are underway.
Remdesivir the best option?
Being a broad-spectrum antiviral agent, Remdesivir becomes an ideal option to turn to when an outbreak involving several unknowns concerning the pathogens involved. The drug was found to exhibit a strong antiviral response against SARS and MERS. Remdesivir was also found to block RNA polymerase of the Ebola virus, thereby preventing its replication.
In February, Chinese authorities announced that they were testing the drug against COVID-19 in trials. Some in vitro studies have highlighted Remdesivir's effectiveness against clinical isolates derived from SARS-CoV-2. Also, the drug was found to be more effective than lopinavir/ritonavir in conjunction with interferon beta according to experimental data from studies associated with the MERS virus in mice.
"Remdesivir has quite high efficacy across all different coronaviruses and therefore it is one of the prime candidates to start being tested," said Munster.









