A company in California's San Rafael, Parnell Pharmaceuticals has developed a nasal spray to prevent Coronavirus infection. Now the developers seek approval from the US Food and Drug Administration (FDA) to market the product.
The company is seeking the FDA's approval to conduct clinical trials on large groups. Meanwhile, Parnell Pharmaceuticals has asked for emergency use authorization. The company will have a meeting with the FDA in December.
Dr. Frank Parnell, a retired nose and throat surgeon who co-founded the company with his wife who is a dermatologist said, "We've not actively promoted it yet. We did make pilot batches. We have some people using it." Both of them graduated from the Georgetown University School of Medicine and completed residency training at the University of Wisconsin at Madison.
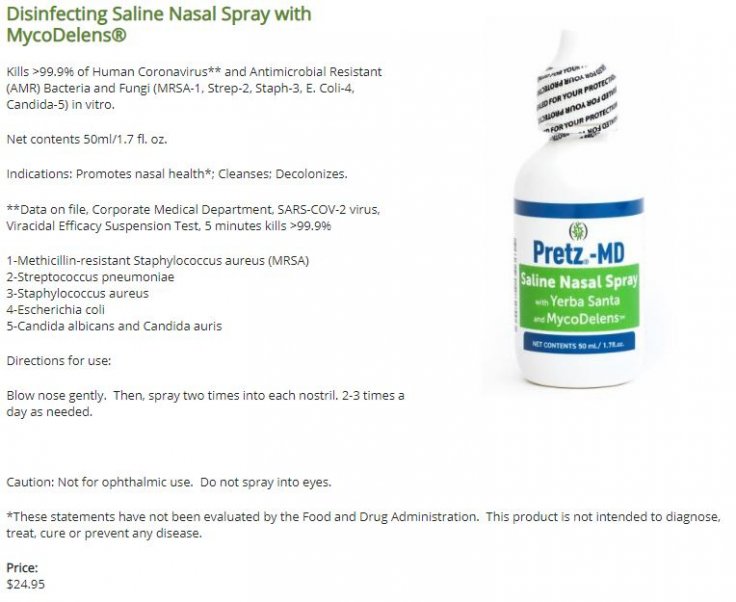
Pretz-MD

As per the developers, the major ingredient in the nasal spray is pentetic acid, which is used in over 150 cosmetic products. It has been approved by the FDA for use by nebulizer or intravenous injection as a treatment for those who have been contaminated by the radioactive plutonium, curium, or americium.
The company which developed the nasal spray said that it must get FDA approval before claiming any effectiveness of the product as a preventive for the viral infection. However, the company has already started selling the spray on its website under the name Pretz-MD.
The product also contains yerba santa, which is generally used for respiratory conditions including cough, cold, tuberculosis, asthma, and long-term swelling of the airways in the lungs. The company has used this ingredient in another product called Pretz Spray, which is a moisturizing nasal spray and has been approved by the FDA as a "class I" medical device.
99.9 Percent Coronavirus Killer
On the website, Parnell says that the Pretz-MD can kill 99.9 percent of the human Coronavirus and anti-microbial resistance (AMR) bacteria, as well as fungi such as MRSA-1 in vitro.
Dr. Lisa Santora, who is the deputy public health officer at Marin County, said that while there is increasing interest in the use of nasal sprays in terms of protection against the Coronavirus, there is not much evidence regarding the safety and efficacy of the Pretz-MD.
"While there is some biological plausibility, there isn't clear evidence yet. We really rely upon the FDA providing its approval and clearance. This product does not seem to be there yet," said Santora.