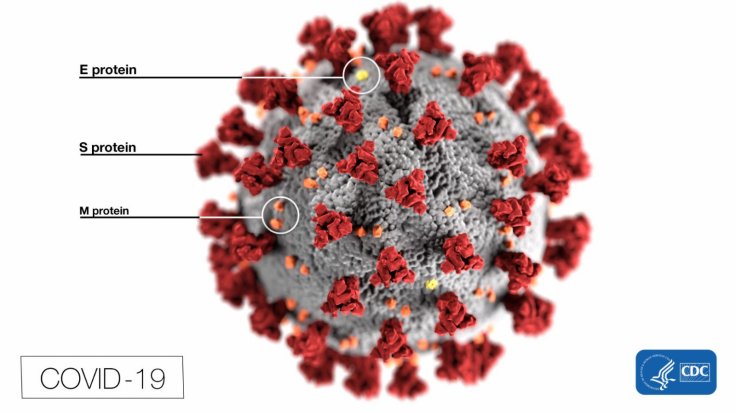
With the number of COVID-19 cases seeing an exponential rise worldwide, an ideal antiviral to treat the coronavirus infection is the need of the hour. Several potential existing drugs are being tested for their effectiveness against the disease. Two such drugs being lopinavir/ritonavir and Arbidol. However, a new study says that they may not be effective options.
According to researchers from Guangzhou Eighth People's Hospital in China, neither of the two drugs improved clinical outcomes in patients suffering from a mild-to-moderate case of the disease and had adverse effects in some.
"We found that neither lopinavir/ritonavir nor Arbidol could benefit clinical outcomes for patients and that they might bring some side effects," said Linghua Li, co-senior author of the study, in a statement.

Lopinavir/ritonavir and Arbidol
Lopinavir/ritonavir(LPV/r) is an antiviral that is a combination of two drugs that are used to treat individuals infected with HIV (human immunodeficiency virus). The combination is often sold under the brand name Kaletra.
Arbidol is the trade name under which Umifenovir is sold. It is an antiviral that is used to treat influenza, especially in China and Russia.
Assigning patients to three groups
86 patients—40 men and 46 women between the ages of 18-80—with a mild-to-moderate manifestation of the disease were examined for the study. They were randomly assigned to receive either of the two antiviral treatments or no antiviral medication at all. 35 received Arbidol, 34 were assigned to the LPV/r set, and antivirals were not administered to 17 patients who formed the control group.
The patients in the LPV/r group received doses of both the drugs twice a day for 7-14 days. For the same duration, the Arbidol group were given the influenza drug three times a day. All three groups were monitored for 21 days, along with supportive care, and oxygen therapy if required.
Similar outcomes
In the study, the authors noted that outcomes in all the three groups were similar at 7 and 14 days. No significant differences in the rates of cough alleviation, reduction of fever, or improved chest CT scan results, were observed.
The trial was also not without adverse events. 12 and 5 patients from the LPV/r and Arbidol groups respectively, experienced severe side effects such as loss of appetite, nausea, and diarrhea during the follow-up period. Nevertheless, no such effects were seen among patients in the control group. "Our findings suggest that we need to cautiously consider before using these drugs," said Li.
Consistent with recent studies
While several countries across the world are looking at HIV drugs as a possible treatment option, the current study joins other recent papers that cast doubts on their effectiveness.
A recently published Chinese study, based on a trial involving 199 patients in Wuhan, found that these HIV drugs did little to improve the conditions of those who were treated with it. Referring to this study, the authors wrote, "The results in our study are consistent with findings from a recent clinical trial of LPV/r in adults hospitalized with severe COVID-19 conducted in Wuhan."
Another retrospective study by Shanghai Public Health Clinical Center in February found that neither LPV/r nor arbidol were effective in improving symptoms or accelerating the reduction of viral load in the 134 patients involved in the trial.
Limitations of the study
In spite of adding to the body of research surrounding the subject, the authors admit that the study is not without its limitations. For starters, the sample size was small.
Also, the patients enrolled were not critically ill and did not include those who had comorbidities that compounded their risks. And finally, the authors also acknowledged that the study was not entirely blinded, which possibly affected its outcome.
Nevertheless, the authors expressed that their research can inform future research. "As a prospective randomized, controlled trial, this study could still provide meaningful suggestions for proper application of LPV/r and arbidol in the treatment of COVID-19," they wrote.








